BrdU-NeuN immunofluorescence
Using
combined BrdU and NeuN labeling, we observed new cells with neuronal
characteristics in the neocortex of adult macaques and rats. Although
some studies have corroborated these findings, others have been unable
to replicate the results. In the case of studies that produced positive
evidence, the number of BrdU-NeuN positive cells reported in the
neocortex is proportionately small relative to the dentate gyrus. When
searching for evidence of new neurons in brain regions where their
occurrence is likely to be relatively rare, sensitive methods must be
used to minimize the likelihood of producing false negative results.
Protocols for labeling tissue with BrdU and NeuN vary widely, and
different approaches to this method may explain some of these
discrepant results. Below we compare the protocols used to label brain
tissue with BrdU and NeuN and provide quantitative analysis of the
signal to noise ratio for the two commonly used methods. We find strong
evidence that the protocol used to stain tissue for BrdU and NeuN in
the studies which report no neocortical neurogenesis in the adult brain
is associated with significantly diminished NeuN signal in the
neocortex of rats and macaques.
Available BrdU
antibodies recognize BrdU in single stranded DNA. Thus, the tissue must
be subjected to denaturation steps before antibody-antigen binding can
occur. These treatments vary greatly from protocol to protocol (see Wojtowicz,
Nature Protocols) – denaturation steps are damaging to tissue
and must be optimized to maximize histological quality. These steps
range from a single 30 min pretreatment at room temperature in HCl to
multiple steps in addition to HCl, such as incubation in heated
formamide (2 hr at 65ºC) and boric acid (pH 8.5). In addition, the
latter protocol is often combined with tissue slicing techniques which
involve freezing.
To examine the
possibility that different protocols produce variations in histological
quality, we processed tissue from 4 rat and 3 monkey brains using the
following two protocols, described below in detail: A) the protocol we
have used to identify BrdU-NeuN positive cells in neocortex. This
method involves minimal pretreatment and no freezing of the tissue; and
B) the protocol used in studies which report no BrdU-NeuN positive
cells in the adult neocortex (Kornack
and Rakic, 2001; Bhadwarj
et al., 2006 – each cite Kuhn
et al., 1996 or a related paper from the same group for
methodological details). The latter protocol differs from our method in
that it involves freezing the brain tissue, as well as treating it with
additional chemicals, including 50% formamide, which has solvent-like
properties, and alkaline borate buffer.
For quantitative analysis,
rat tissue was stained using protocol A and protocol B, as well as
protocol B without freezing the tissue (the effects
of freezing tissue on staining using Protocol A were not tested).
After reactions were complete, scans from the anterior neocortex of the
rat were made using a Zeiss Axiovert confocal microscope with an Argon
laser and 510LSM software. Data analysis proceeded as
follows: One µm-thick optical sections
were scanned from the anterior neocortex; 10 frames (115.16µm2) were obtained
per animal using each protocol, holding the scanning parameters
constant. Outlines of all NeuN stained cell bodies in the scans were
traced using the measurement tool in the LSM software and the mean
intensity for each cell body was determined (0-255 grayscale). The
outline was then moved to an adjacent region, which did not contain any
NeuN positive cell bodies, and a background optical intensity measure
of the same size area was recorded. Every labeled cell was analyzed in
this manner. The signal was defined as the mean intensity value for the
cell bodies. The background was defined as the mean intensity value for
the comparably sized regions containing no stained cell bodies. Signal
to noise ratio was calculated according to the following formula:
(Signal – Background) / SD
Background
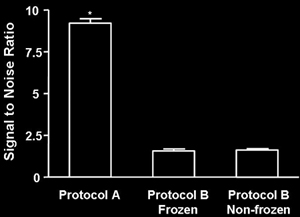
We found that tissue
stained for NeuN using Protocol A had a substantially higher signal to
noise ratio than tissue stained for NeuN using protocol B. Freezing the
tissue did not appear to have any measurable effect on NeuN staining in
tissue stained with Protocol B. Data were analyzed using
one-way analysis of variance (F(2, 590) = 352, p < 0.0001; Tukey
post hoc, p < 0.001 for both Protocol A versus B comparisons).
Examples of images of NeuN
staining taken from rat tissue used in this analysis can be previewed
as jpegs under Images . Additional examples
of monkey tissue stained
with these protocols are also provided.
Methods
PROTOCOL A – for
40µm-thick sections from non-frozen brain tissue.
Day 1
1). Incubate in 2N HCl:TBS
for 30min
2). Incubate in rat
anti-BrdU:TBS (1:200; Accurate) with mouse anti-NeuN (1:500; Chemicon)
and 25 µl/ml of 10% Tween-20 overnight at 4ºC
Note: all reactions take
place at room temperature unless otherwise noted; each incubation is
preceded by 3 rinses in TBS (3-5 min each).
Day 2
1). Incubate in
biotinylated anti-rat:TBS (1:250; Vector) for 90 min
2). Incubate in
Streptavidin Alexa 568:TBS (1:1000; Molecular Probes) for 1 hr in the
dark
3). Incubate in
secondary goat anti-mouse Alexa 488:TBS (1:500; Molecular Probes) for
30 min in the dark
4). Rinse, mount,
dry in the dark, coverslip under 75% Glycerol:TBS
PROTOCOL B – for
frozen brain tissue (first cryoprotected in 30% sucrose).
Day 1
1). Incubate in 50%
formamide/2xSSC (0.3M NaCl, 0.03M sodium citrate) for 2 hr at 65ºC
2). Rinse in 2xSSC for 5
min
3). Incubate in 2N HCl at
37ºC for 30 min
4). Rinse in 0.1M boric
acid (pH 8.5) for 10 min
5). Rinse 3x in TBS
6). Incubate in TBS/0.25%
Triton X-100/3% normal horse serum (TBS-TS) for 30 min
7). Incubate in rat
anti-BrdU:TBS-TS (1:200; Accurate) with mouse anti-NeuN (1:500;
Chemicon) overnight at 4ºC
Day 2
Same as Protocol A
|




